A Broad Suite of AI Tools
for Chest Conditions
From pulmonary embolism to aortic dissection, we provide AI solutions that can support radiology departments in the detection and treatment of highly morbid thoraco-abdominal conditions. Our tools excel in overcoming the challenges associated with the diversity of chest conditions, non-specific indicators, and the potential overlap of symptoms with other diseases.
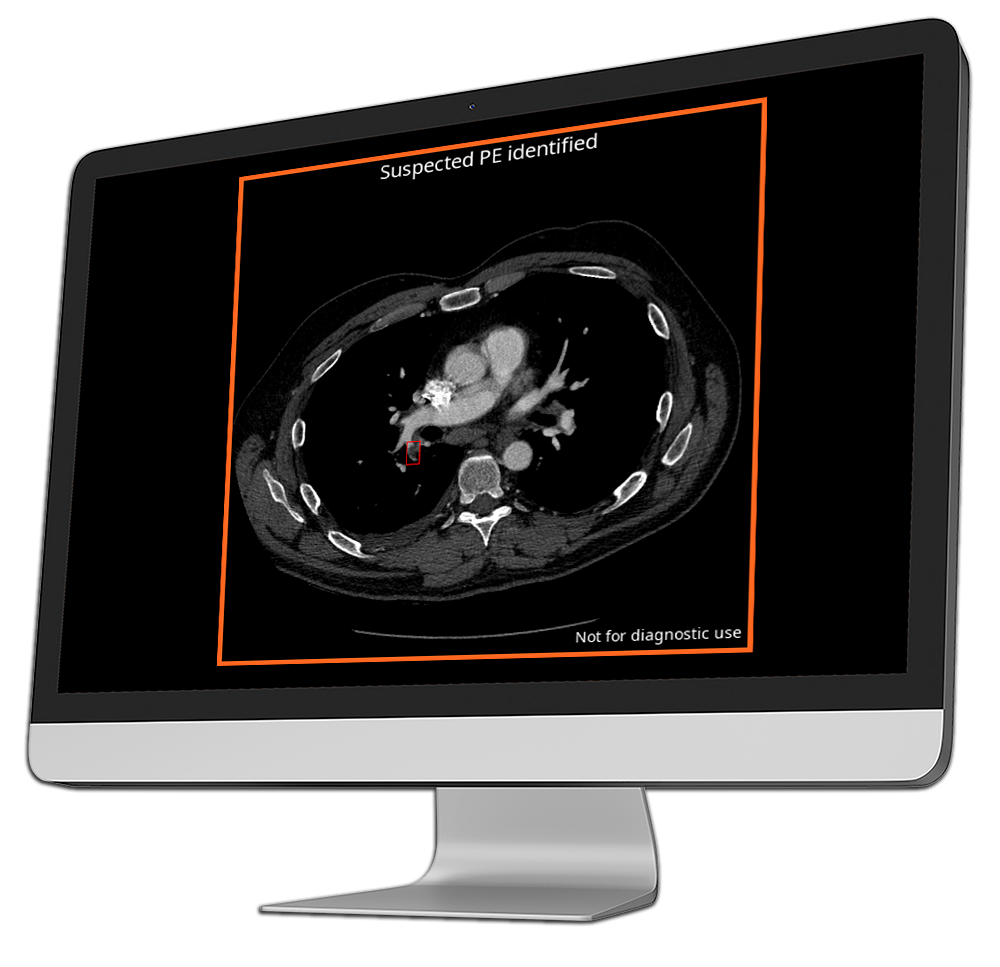
CINA-PE
Pulmonary Embolism
Pulmonary embolism (PE) is a major global health concern, causing significant morbidity, mortality, and hospitalizations worldwide.
CINA-PE is an AI-powered triage tool that automatically analyzes chest CT scans to detect pulmonary embolisms, helping identify life-threatening blockages in the pulmonary arteries for faster emergency decision-making support.
By rapidly flagging these critical cases, CINA-PE enables radiologists to prioritize urgent patients, reduce diagnosis time, and accelerate access to treatment to improve patient outcomes.
Main Benefits
- Prioritizes critical cases.
- Accelerates clinical decision-making.
- Enhances care coordination by providing fast, reliable results to the clinical team.
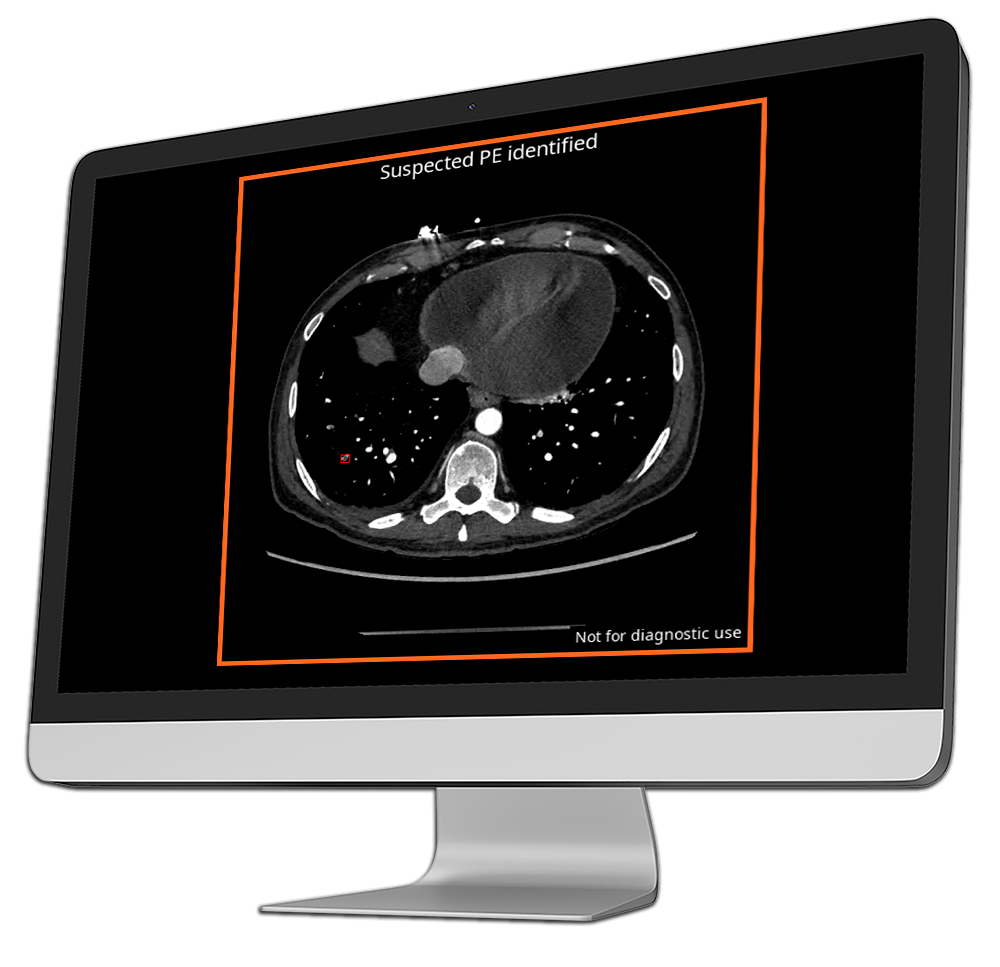
CINA-iPE
Incidental Pulmonary Embolism
In diagnostic imaging, incidental findings such as pulmonary embolisms (PE) are often overlooked, creating risks for delayed treatment and severe complications.
CINA-iPE is an AI-powered triage tool that detects unsuspected pulmonary embolisms on CT scans performed for other health conditions that include the lung region—such as chest, abdomen-pelvis, thoraco-abdominal, or whole-body scans.
By flagging unexpected PE that might otherwise go unnoticed, CINA-iPE enables radiologists to initiate early treatment, preventing missed diagnoses, emergency readmissions, and the associated health, time, and cost burdens for both patients and hospitals.
Main Benefits
- Prevents missed diagnoses by detecting opportunistic pulmonary embolism
- Enables early treatment to reduce the risk of severe complications and emergency readmissions
- Improves efficiency and reduces costs by avoiding delayed care that impacts both patients and hospitals.
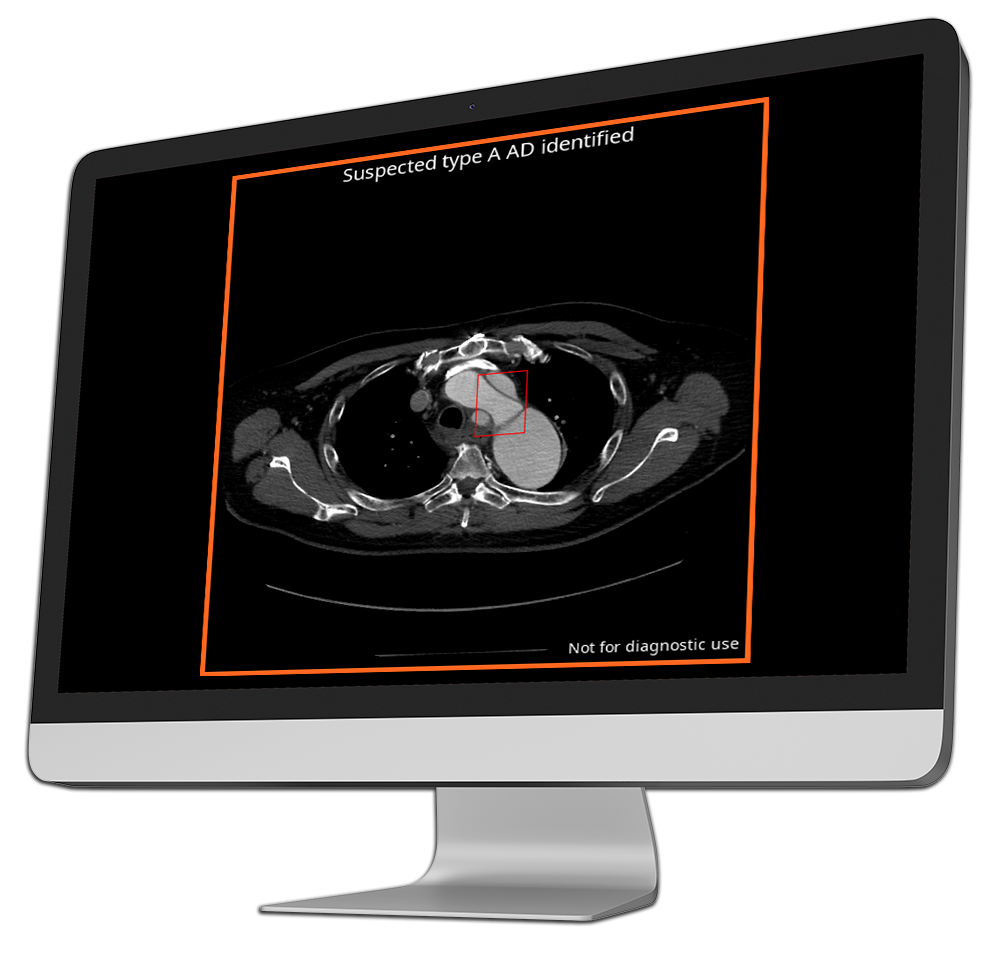
CINA-AD
Aortic dissection
Aortic dissection is a rare but life-threatening condition where a tear in the aorta can rapidly lead to catastrophic outcomes if not treated promptly.
CINA-AD is an AI-powered triage tool that detects acute type A and type B aortic dissections on chest or thoracoabdominal CT angiography, flagging critical cases in real time.
By enabling radiologists to quickly prioritize patients for emergency intervention, CINA-AD reduces the risk of delayed diagnosis, supports faster decision-making, and improves survival in this highly time-sensitive condition.
Main Benefits
- Enables early detection of life-threatening aortic dissections to support timely emergency intervention.
- Helps radiologists prioritize critical patients and reduce the risk of delayed diagnosis.
- Improves survival chances in one of the most time-sensitive vascular emergencies.
CINA-PE, CINA-AD and CINA-iPE, medical images analysis softwares, are medical devices manufactured by Avicenna.AI. These medical devices are reserved for health professionals. The performance and outputs of these tools may differ slightly according to the software version and applicable regulations.
These software have been designed and manufactured according to the EN ISO 13485 Quality management system. Read the instructions in the notice carefully before any use.
- Instructions for Use are available here.
- Manufacturer: Avicenna.AI (France).
- Medical devices Class II following the Code of Federal Regulations of the United States of America 21CFR on Medical Devices.
- Medical devices Class IIb following the European Regulation on Medical Device (EU) 2017/745 .
CINA Chest
Download our Case study
Complete the form below to download our Case Study.
